Glass Delamination Analysis Services Lab
Watch our Glass Delamination Analysis Webinar
- Glass delamination analysis
- Validation and testing of product glass containers
- USP <1660> testing support
In the pharmaceutical industry, glass delamination is defined as thin glass flakes shedding from the internal diameter of glass containers. The flakes, also called “glass lamellae,” can be shed and suspended in parenteral drug products. This is a major concern in the pharmaceutical industry and has resulted in a number of recalls. Detecting glass lamellae is one of the key aspects of glass delamination analysis. USP laid out some analytical guidelines for pharmaceutical manufacturers and analytical laboratories on how to screen for glass delamination.
Examination of internal diameter of glass container
USP <1660> recommends three key parameters for screening glass delamination in glass containers. The parameters are the condition of the glass surface as received, the presence of extracted elements in solution, as well as the presence of visible and subvisible glass particles.
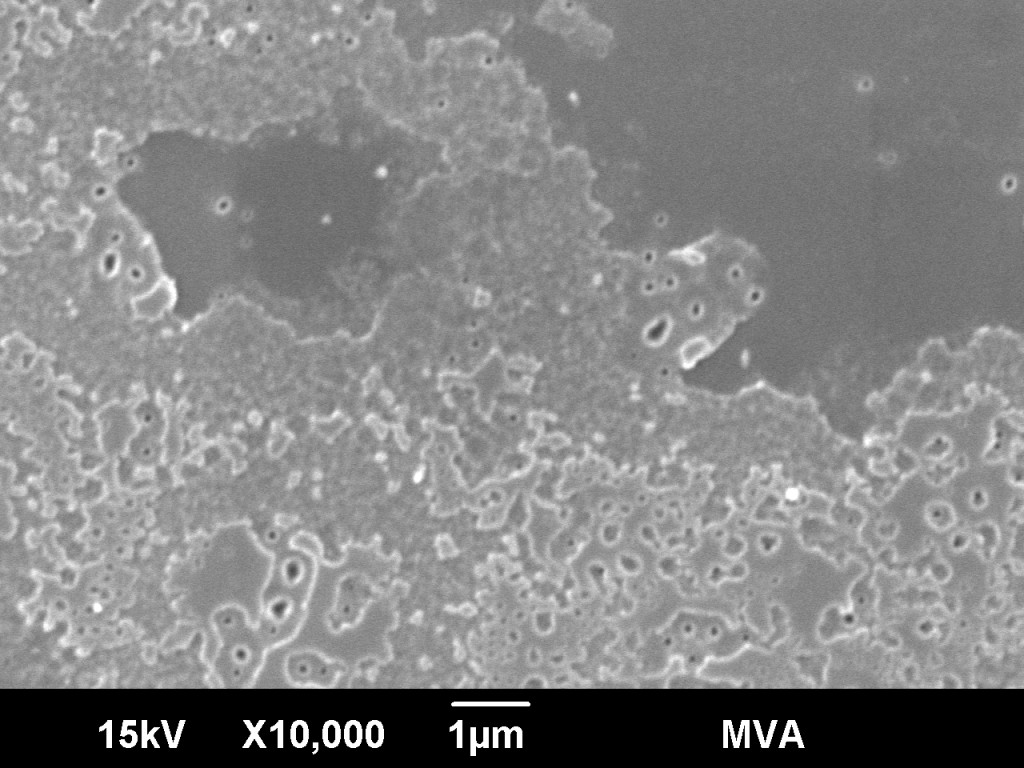
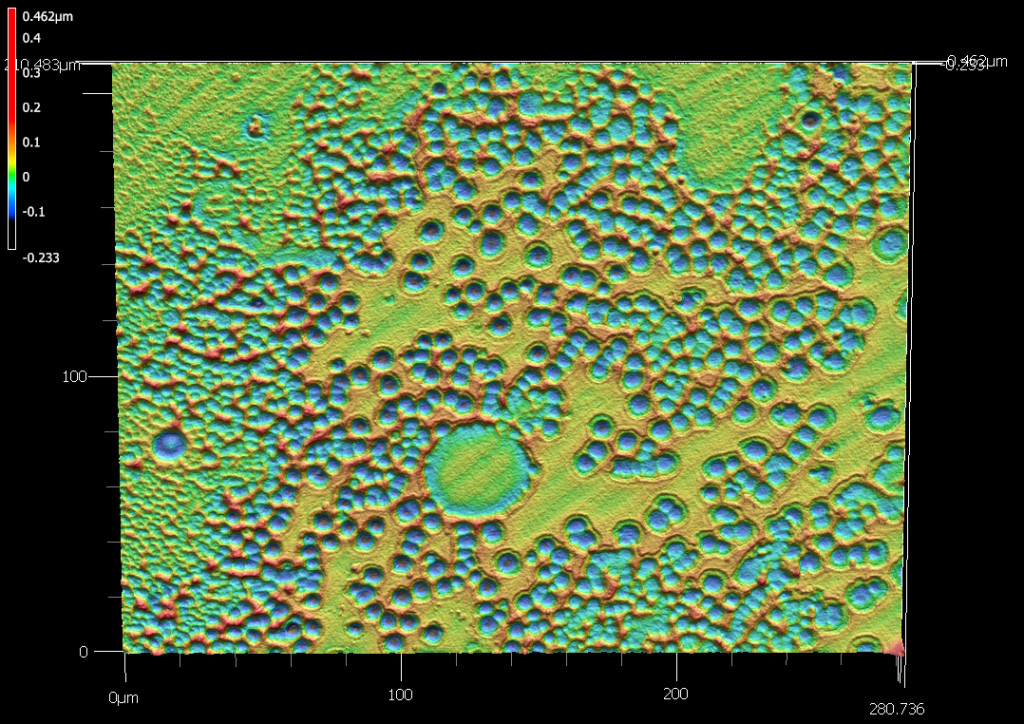
- For glass surface analysis, the objective is to examine the degree of surface pitting and chemical composition as a function of depth. This part of the analysis can be accomplished by differential interference microscopy or electron microscopy and secondary ion mass spectrometry.
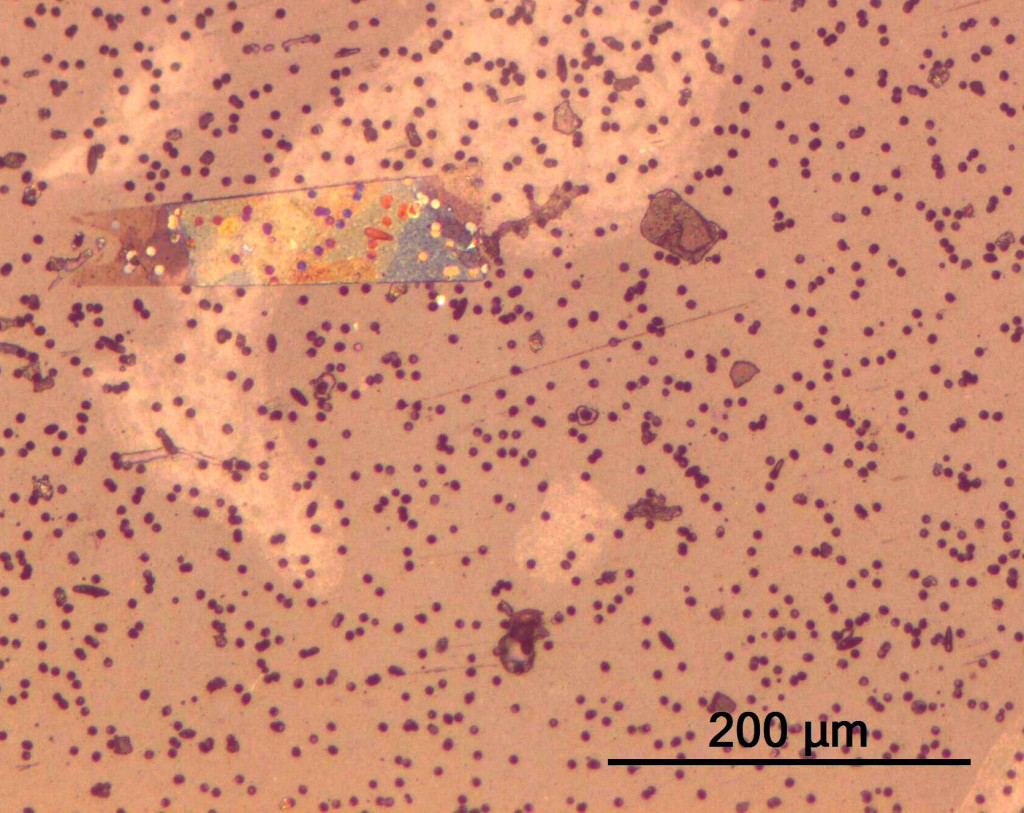
- For visible and subvisible glass particles, the objective is to examine and record the number, size, shape and composition of the particles. This part of the study can be accomplished by a particle size analyzer or SEM-EDS.

Analytical approach for examination of glass container
MVA Scientific Consultants has customized a systematic approach for glass delamination analysis which includes special sample preparation, filtration, and examination by light microscopy and SEM-EDS.
-
- Glass container examination– upon receiving the samples, the first step is to examine
- the internal diameter of the glass containers by stereomicroscopy and differential interference contrast (DIC) to document any visible defects.
-
- Remove and filter content – re-examine internal diameter of the containers by stereomicroscopy and differential interference contrast (DIC) and examine the filter for lamella by DIC and SEM-EDS.
-
- Glass container preparation – glass containers come in all shapes and sizes; they need to be sectioned to expose the interior surface for analysis.
-
- Microscopical inspection of sectioned containers – re-examine, document and photograph the interior surface of the glass containers by DIC.
Analysis of sectioned containers by SEM – analyze the sectioned glass container by SEM-EDS for surface delamination.



For more info visit:
Looking at Glass Delamination of Drug Vials
Email us or call us at 770-662-8509 to learn more about how we can help you with glass delamination analysis.




